In physics (in particular in statistical mechanics), the Maxwell–Boltzmann distribution, or Maxwell(ian) distribution, is a particular probability distribution named after James Clerk Maxwell and Ludwig Boltzmann.
Probability density function  | |||
Cumulative distribution function  | |||
| Parameters | |||
|---|---|---|---|
| Support | |||
(where exp is the exponential function) | |||
| CDF | (where erf is the error function) | ||
| Mean | |||
| Mode | |||
| Variance | |||
| Skewness | |||
| Excess kurtosis | |||
| Entropy | |||
It was first defined and used for describing particle speeds in idealized gases, where the particles move freely inside a stationary container without interacting with one another, except for very brief collisions in which they exchange energy and momentum with each other or with their thermal environment. The term "particle" in this context refers to gaseous particles only (atoms or molecules), and the system of particles is assumed to have reached thermodynamic equilibrium.[1] The energies of such particles follow what is known as Maxwell–Boltzmann statistics, and the statistical distribution of speeds is derived by equating particle energies with kinetic energy.
Mathematically, the Maxwell–Boltzmann distribution is the chi distribution with three degrees of freedom (the components of the velocity vector in Euclidean space), with a scale parameter measuring speeds in units proportional to the square root of (the ratio of temperature and particle mass).[2]
The Maxwell–Boltzmann distribution is a result of the kinetic theory of gases, which provides a simplified explanation of many fundamental gaseous properties, including pressure and diffusion.[3] The Maxwell–Boltzmann distribution applies fundamentally to particle velocities in three dimensions, but turns out to depend only on the speed (the magnitude of the velocity) of the particles. A particle speed probability distribution indicates which speeds are more likely: a randomly chosen particle will have a speed selected randomly from the distribution, and is more likely to be within one range of speeds than another. The kinetic theory of gases applies to the classical ideal gas, which is an idealization of real gases. In real gases, there are various effects (e.g., van der Waals interactions, vortical flow, relativistic speed limits, and quantum exchange interactions) that can make their speed distribution different from the Maxwell–Boltzmann form. However, rarefied gases at ordinary temperatures behave very nearly like an ideal gas and the Maxwell speed distribution is an excellent approximation for such gases. This is also true for ideal plasmas, which are ionized gases of sufficiently low density.[4]
The distribution was first derived by Maxwell in 1860 on heuristic grounds.[5] Boltzmann later, in the 1870s, carried out significant investigations into the physical origins of this distribution. The distribution can be derived on the ground that it maximizes the entropy of the system. A list of derivations are:
- Maximum entropy probability distribution in the phase space, with the constraint of conservation of average energy
- Canonical ensemble.
Distribution function
For a system containing a large number of identical non-interacting, non-relativistic classical particles in thermodynamic equilibrium, the fraction of the particles within an infinitesimal element of the three-dimensional velocity space d 3v, centered on a velocity vector of magnitude v, is given by

- m is the particle mass;
- k is the Boltzmann constant;
- T is thermodynamic temperature;
- f (v) is a probability distribution function, properly normalized so that
over all velocities is unity.
One can write the element of velocity space as 



The Maxwellian distribution function for particles moving in only one direction, if this direction is x, is

Recognizing the symmetry of 

This probability density function gives the probability, per unit speed, of finding the particle with a speed near v. This equation is simply the Maxwell–Boltzmann distribution (given in the infobox) with distribution parameter 

The simplest ordinary differential equation satisfied by the distribution is:

or in unitless presentation:


Relaxation to the 2D Maxwell–Boltzmann distribution
For particles confined to move in a plane, the speed distribution is given by

This distribution is used for describing systems in equilibrium. However, most systems do not start out in their equilibrium state. The evolution of a system towards its equilibrium state is governed by the Boltzmann equation. The equation predicts that for short range interactions, the equilibrium velocity distribution will follow a Maxwell–Boltzmann distribution. To the right is a molecular dynamics (MD) simulation in which 900 hard sphere particles are constrained to move in a rectangle. They interact via perfectly elastic collisions. The system is initialized out of equilibrium, but the velocity distribution (in blue) quickly converges to the 2D Maxwell–Boltzmann distribution (in orange).
Typical speeds
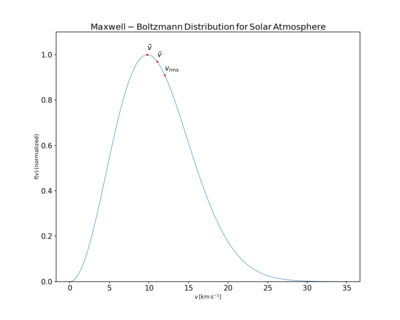
 ,
,  , and Vrms mark the most probable, mean, and root mean square velocities, respectively. Their values are
, and Vrms mark the most probable, mean, and root mean square velocities, respectively. Their values are  ≈ 9.79 km/s,
≈ 9.79 km/s,  ≈ 11.05 km/s, and Vrms ≈ 12.00 km/s.
≈ 11.05 km/s, and Vrms ≈ 12.00 km/s.The mean speed 

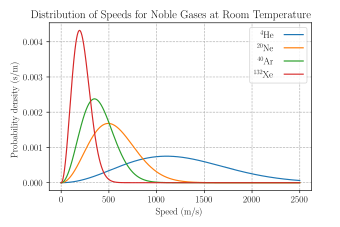
This works well for nearly ideal, monatomic gases like helium, but also for molecular gases like diatomic oxygen. This is because despite the larger heat capacity (larger internal energy at the same temperature) due to their larger number of degrees of freedom, their translational kinetic energy (and thus their speed) is unchanged.[7]
- The most probable speed, vp, is the speed most likely to be possessed by any molecule (of the same mass m) in the system and corresponds to the maximum value or the mode of f(v). To find it, we calculate the derivative
set it to zero and solve for v:
with the solution:where:- R is the gas constant;
- M is molar mass of the substance, and thus may be calculated as a product of particle mass, m, and Avogadro constant, NA:
For diatomic nitrogen (N2, the primary component of air)[8] at room temperature (300 K), this gives

- The mean speed is the expected value of the speed distribution, setting
:
- The mean square speed
is the second-order raw moment of the speed distribution. The "root mean square speed"
is the square root of the mean square speed, corresponding to the speed of a particle with average kinetic energy, setting
:

In summary, the typical speeds are related as follows:

The root mean square speed is directly related to the speed of sound c in the gas, by

 is the adiabatic index, f is the number of degrees of freedom of the individual gas molecule. For the example above, diatomic nitrogen (approximating air) at 300 K,
is the adiabatic index, f is the number of degrees of freedom of the individual gas molecule. For the example above, diatomic nitrogen (approximating air) at 300 K,  [9] and
[9] and
The average relative velocity


The integral can easily be done by changing to coordinates 

Limitations
The Maxwell–Boltzmann distribution assumes that the velocities of individual particles are much less than the speed of light, i.e. that 

Maxwell–Boltzmann statistics
The original derivation in 1860 by James Clerk Maxwell was an argument based on molecular collisions of the Kinetic theory of gases as well as certain symmetries in the speed distribution function; Maxwell also gave an early argument that these molecular collisions entail a tendency towards equilibrium.[5][10] After Maxwell, Ludwig Boltzmann in 1872[11] also derived the distribution on mechanical grounds and argued that gases should over time tend toward this distribution, due to collisions (see H-theorem). He later (1877)[12] derived the distribution again under the framework of statistical thermodynamics. The derivations in this section are along the lines of Boltzmann's 1877 derivation, starting with result known as Maxwell–Boltzmann statistics (from statistical thermodynamics). Maxwell–Boltzmann statistics gives the average number of particles found in a given single-particle microstate. Under certain assumptions, the logarithm of the fraction of particles in a given microstate is linear in the ratio of the energy of that state to the temperature of the system: there are constants 



This relation can be written as an equation by introducing a normalizing factor:
where:
- Ni is the expected number of particles in the single-particle microstate i,
- N is the total number of particles in the system,
- Ei is the energy of microstate i,
- the sum over index j takes into account all microstates,
- T is the equilibrium temperature of the system,
- k is the Boltzmann constant.
The denominator in Equation (1) is a normalizing factor so that the ratios 
Because velocity and speed are related to energy, Equation (1) can be used to derive relationships between temperature and the speeds of gas particles. All that is needed is to discover the density of microstates in energy, which is determined by dividing up momentum space into equal sized regions.
Distribution for the momentum vector
The potential energy is taken to be zero, so that all energy is in the form of kinetic energy.The relationship between kinetic energy and momentum for massive non-relativistic particles is
where p2 is the square of the momentum vector p = [px, py, pz]. We may therefore rewrite Equation (1) as:
where:
- Z is the partition function, corresponding to the denominator in Equation (1);
- m is the molecular mass of the gas;
- T is the thermodynamic temperature;
- k is the Boltzmann constant.
This distribution of Ni : N is proportional to the probability density function fp for finding a molecule with these values of momentum components, so:






























































