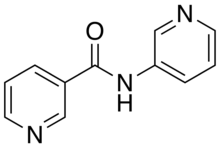
The organic compound 3-pyridylnicotinamide (3-pna), also known as N-(pyridin-3-yl)nicotinamide, is a kinked dipodal dipyridine that is synthesized through the reaction of nicotinoyl chloride and 3-aminopyridine.[1] The nitrogen atoms on its pyridine rings, like those of its isomer 4-pyridylnicotinamide, can donate their electron lone pairs to metal cations, allowing it to bridge metal centers and act as a bidentate ligand in coordination polymers.[2][3][4][5][6] It can be used to synthesize polymers that have potentially useful gas adsorption properties.[2][3]
 | |
| Names | |
|---|---|
| Preferred IUPAC name N-(Pyridin-3-yl)pyridine-3-carboxamide | |
| Other names 3-pna 3-pyridinecarboxamide, N-3-pyridinyl- N-(Pyridin-3-yl)nicotinamide | |
| Identifiers | |
3D model (JSmol) | |
| ChEMBL | |
| ChemSpider | |
PubChem CID | |
| UNII | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| C11H9N3O | |
| Molar mass | 199.20 g/mol |
| Density | 1.287 g/cm3 |
| Boiling point | 286.08 °C (546.94 °F; 559.23 K) |
| Structure | |
| 0 D | |
| Hazards | |
| Flash point | 127 °C (261 °F; 400 K) |
| Related compounds | |
Related compounds | 4,4'-bipyridine Pyridine Nicotinamide 3-Aminopyridine 4-Pyridylnicotinamide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |