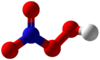
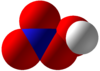
Peroxynitric acid or peroxonitric acid is a chemical compound with the formula HNO
4. It is an oxyacid of nitrogen, after peroxynitrous acid.
 | |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name Hydroxy nitrate | |||
| Systematic IUPAC name | |||
| Identifiers | |||
3D model (JSmol) | |||
| ChemSpider | |||
PubChem CID | |||
CompTox Dashboard (EPA) | |||
| |||
| Properties | |||
| HNO4 | |||
| Molar mass | 79.01224 g/mol | ||
| Conjugate base | Peroxynitrate | ||
| Related compounds | |||
Related compounds | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
Preparation
Peroxynitrate, the conjugate base of peroxynitric acid, is formed rapidly during decomposition of peroxynitrite in neutral conditions.[4]
Atmospheric chemistry
Peroxynitric acid is formed in the atmosphere, although it is unstable, it is important as a reservoir for NO2 through the reversible radical reaction:[5]
- HO
2NO
2 ⇌ HO•
2 + NO•
2