Phosphate
salts or esters of phosphoric acid
A phosphate is a salt of phosphoric acid. Phosphates are important in biochemistry.Phosphates have the formula PO43- and a molar mass of 94.973 g/mol. An example of a phosphate is sodium phosphate. Three different types of phosphates are known. They are orthophosphate, PO43-; metaphosphate, PO32-; and pyrophosphate, P2O73-. They have a combining power of 3.
| |||
| Names | |||
|---|---|---|---|
| Systematic IUPAC name Phosphate[1] | |||
| Identifiers | |||
| |||
3D model (JSmol) | |||
| Beilstein Reference | 3903772 | ||
| ChEBI | |||
| ChemSpider | |||
| Gmelin Reference | 1997 | ||
| MeSH | Phosphates | ||
PubChem CID | |||
| UNII | |||
SMILES
| |||
| Properties | |||
| PO3− 4 | |||
| Molar mass | 94.9714 g mol−1 | ||
| Conjugate acid | Hydrogen phosphate | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||

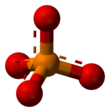
Structure
Phosphates are made of one phosphorus atom surrounded by four oxygen atoms. Many phosphates do not dissolve in water.
Sources
🔥 Top keywords: Main PageSpecial:Search0Slash (punctuation)BlackSpecial:RecentChanges4 (number)DavidSOLID (object-oriented design)Wikipedia:AboutFile:Sexual intercourse with internal ejaculation.webmHelp:ContentsHelp:IntroductionLisa Sparxxx2023 UEFA Champions League FinalColour24-hour clockAdolf Hitler UunonaBismillahir Rahmanir Raheem6 (number)T. N. SeshanFile:ASCII-Table-wide.svg20 (number)Poor Things (movie)United StatesCristiano RonaldoList of people who have walked on the MoonAli Malikov50 (number)17 (number)The Valley (2024 TV series)GrassList of mathematical symbolsList of U.S. states and territories by time zone8 (number)List of countries by areaWikipedia:Simple talkList of largest Hindu templesRama