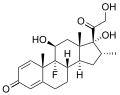
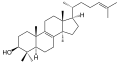
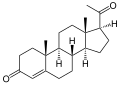
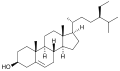
A steroid is an organic compound with four fused rings (designated A, B, C, and D) arranged in a specific molecular configuration.

Steroids have two principal biological functions: as important components of cell membranes that alter membrane fluidity; and as signaling molecules. Examples include the lipid cholesterol, sex hormones estradiol and testosterone,[2]: 10–19 anabolic steroids, and the anti-inflammatory corticosteroid drug dexamethasone.[3] Hundreds of steroids are found in fungi, plants, and animals. All steroids are manufactured in cells from the sterols lanosterol (opisthokonts) or cycloartenol (plants). Lanosterol and cycloartenol are derived from the cyclization of the triterpene squalene.[4]
Steroids are named after the steroid cholesterol[5] which was first described in gall stones from Ancient Greek chole- 'bile' and stereos 'solid'.[6][7][8]
The steroid nucleus (core structure) is called gonane (cyclopentanoperhydrophenanthrene).[9] It is typically composed of seventeen carbon atoms, bonded in four fused rings: three six-member cyclohexane rings (rings A, B and C in the first illustration) and one five-member cyclopentane ring (the D ring). Steroids vary by the functional groups attached to this four-ring core and by the oxidation state of the rings. Sterols are forms of steroids with a hydroxy group at position three and a skeleton derived from cholestane.[1]: 1785f [10] Steroids can also be more radically modified, such as by changes to the ring structure, for example, cutting one of the rings. Cutting Ring B produces secosteroids one of which is vitamin D3.
Nomenclature
Rings and functional groups


Gonane, also known as steran or cyclopentanoperhydrophenanthrene, the simplest steroid and the nucleus of all steroids and sterols,[11][12] is composed of seventeen carbon atoms in carbon-carbon bonds forming four fused rings in a three-dimensional shape. The three cyclohexane rings (A, B, and C in the first illustration) form the skeleton of a perhydro derivative of phenanthrene. The D ring has a cyclopentane structure. When the two methyl groups and eight carbon side chains (at C-17, as shown for cholesterol) are present, the steroid is said to have a cholestane framework. The two common 5α and 5β stereoisomeric forms of steroids exist because of differences in the side of the largely planar ring system where the hydrogen (H) atom at carbon-5 is attached, which results in a change in steroid A-ring conformation. Isomerisation at the C-21 side chain produces a parallel series of compounds, referred to as isosteroids.[13]
Examples of steroid structures are:
- Dexamethasone, a synthetic corticosteroid drug
- Lanosterol, the biosynthetic precursor to animal steroids. The number of carbons (30) indicates its triterpenoid classification.
- Progesterone, a steroid hormone involved in the female menstrual cycle, pregnancy, and embryogenesis
- Medrogestone, a synthetic drug with effects similar to progesterone
- β-Sitosterol, a plant or phytosterol, with a fully branched hydrocarbon side chain at C-17 and an hydroxyl group at C-3
In addition to the ring scissions (cleavages), expansions and contractions (cleavage and reclosing to a larger or smaller rings)—all variations in the carbon-carbon bond framework—steroids can also vary:
- in the bond orders within the rings,
- in the number of methyl groups attached to the ring (and, when present, on the prominent side chain at C17),
- in the functional groups attached to the rings and side chain, and
- in the configuration of groups attached to the rings and chain.[2]: 2–9
For instance, sterols such as cholesterol and lanosterol have a hydroxyl group attached at position C-3, while testosterone and progesterone have a carbonyl (oxo substituent) at C-3. Among these compounds, only lanosterol has two methyl groups at C-4. Cholesterol which has a C-5 to C-6 double bond, differs from testosterone and progesterone which have a C-4 to C-5 double bond.
 |  |
Naming convention
Almost all biologically relevant steroids can be presented as a derivative of a parent cholesterol-like hydrocarbon structure that serves as a skeleton.[14][15] These parent structures have specific names, such as pregnane, androstane, etc. The derivatives carry various functional groups called suffixes or prefixes after the respective numbers, indicating their position in the steroid nucleus.[16] There are widely used trivial steroid names of natural origin with significant biologic activity, such as progesterone, testosterone or cortisol. Some of these names are defined in The Nomenclature of Steroids.[17] These trivial names can also be used as a base to derive new names, however, by adding prefixes only rather than suffixes, e.g., the steroid 17α-hydroxyprogesterone has a hydroxy group (-OH) at position 17 of the steroid nucleus comparing to progesterone.
The letters α and β[18] denote absolute stereochemistry at chiral centers—a specific nomenclature distinct from the R/S convention[19] of organic chemistry to denote absolute configuration of functional groups, known as Cahn–Ingold–Prelog priority rules. The R/S convention assigns priorities to substituents on a chiral center based on their atomic number. The highest priority group is assigned to the atom with the highest atomic number, and the lowest priority group is assigned to the atom with the lowest atomic number. The molecule is then oriented so that the lowest priority group points away from the viewer, and the remaining three groups are arranged in order of decreasing priority around the chiral center. If this arrangement is clockwise, it is assigned an R configuration; if it is counterclockwise, it is assigned an S configuration.[20] In contrast, steroid nomenclature uses α and β to denote stereochemistry at chiral centers. The α and β designations are based on the orientation of substituents relative to each other in a specific ring system. In general, α refers to a substituent that is oriented towards the plane of the ring system, while β refers to a substituent that is oriented away from the plane of the ring system. In steroids drawn from the standard perspective used in this paper, α-bonds are depicted on figures as dashed wedges and β-bonds as solid wedges.[14]
The name "11-deoxycortisol" is an example of a derived name that uses cortisol as a parent structure without an oxygen atom (hence "deoxy") attached to position 11 (as a part of a hydroxy group).[14][21] The numbering of positions of carbon atoms in the steroid nucleus is set in a template found in the Nomenclature of Steroids[22] that is used regardless of whether an atom is present in the steroid in question.[14]
Unsaturated carbons (generally, ones that are part of a double bond) in the steroid nucleus are indicated by changing -ane to -ene.[23] This change was traditionally done in the parent name, adding a prefix to denote the position, with or without Δ (Greek capital delta) which designates unsaturation, for example, 4-pregnene-11β,17α-diol-3,20-dione (also Δ4-pregnene-11β,17α-diol-3,20-dione) or 4-androstene-3,11,17-trione (also Δ4-androstene-3,11,17-trione). However, the Nomenclature of Steroids recommends the locant of a double bond to be always adjacent to the syllable designating the unsaturation, therefore, having it as a suffix rather than a prefix, and without the use of the Δ character, i.e. pregn-4-ene-11β,17α-diol-3,20-dione or androst-4-ene-3,11,17-trione. The double bond is designated by the lower-numbered carbon atom, i.e. "Δ4-" or "4-ene" means the double bond between positions 4 and 5. The saturation of carbons of a parent steroid can be done by adding "dihydro-" prefix,[24] i.e., a saturation of carbons 4 and 5 of testosterone with two hydrogen atoms is 4,5α-dihydrotestosterone or 4,5β-dihydrotestosterone. Generally, when there is no ambiguity, one number of a hydrogen position from a steroid with a saturated bond may be omitted, leaving only the position of the second hydrogen atom, e.g., 5α-dihydrotestosterone or 5β-dihydrotestosterone. The Δ5-steroids are those with a double bond between carbons 5 and 6 and the Δ4 steroids are those with a double bond between carbons 4 and 5.[25][23]
The abbreviations like "P4" for progesterone and "A4" for androstenedione for refer to Δ4-steroids, while "P5" for pregnenolone and "A5" for androstenediol refer to Δ5-steroids.[14]
The suffix -ol denotes a hydroxy group, while the suffix -one denotes an oxo group. When two or three identical groups are attached to the base structure at different positions, the suffix is indicated as -diol or -triol for hydroxy, and -dione or -trione for oxo groups, respectively. For example, 5α-pregnane-3α,17α-diol-20-one has a hydrogen atom at the 5α position (hence the "5α-" prefix), two hydroxy groups (-OH) at the 3α and 17α positions (hence "3α,17α-diol" suffix) and an oxo group (=O) at the position 20 (hence the "20-one" suffix). However, erroneous use of suffixes can be found, e.g., "5α-pregnan-17α-diol-3,11,20-trione"[26] [sic] — since it has just one hydroxy group (at 17α) rather than two, then the suffix should be -ol, rather than -diol, so that the correct name to be "5α-pregnan-17α-ol-3,11,20-trione".
According to the rule set in the Nomenclature of Steroids, the terminal "e" in the parent structure name should be elided before the vowel (the presence or absence of a number does not affect such elision).[14][16] This means, for instance, that if the suffix immediately appended to the parent structure name begins with a vowel, the trailing "e" is removed from that name. An example of such removal is "5α-pregnan-17α-ol-3,20-dione", where the last "e" of "pregnane" is dropped due to the vowel ("o") at the beginning of the suffix -ol. Some authors incorrectly use this rule, eliding the terminal "e" where it should be kept, or vice versa.[27]
The term "11-oxygenated" refers to the presence of an oxygen atom as an oxo (=O) or hydroxy (-OH) substituent at carbon 11. "Oxygenated" is consistently used within the chemistry of the steroids[28] since the 1950s.[29] Some studies use the term "11-oxyandrogens"[30][31] as an abbreviation for 11-oxygenated androgens, to emphasize that they all have an oxygen atom attached to carbon at position 11.[32][33] However, in chemical nomenclature, the prefix "oxy" is associated with ether functional groups, i.e., a compound with an oxygen atom connected to two alkyl or aryl groups (R-O-R),[34] therefore, using "oxy" within the name of a steroid class may be misleading. One can find clear examples of "oxygenated" to refer to a broad class of organic molecules containing a variety of oxygen containing functional groups in other domains of organic chemistry,[35] and it is appropriate to use this convention.[14]
Even though "keto" is a standard prefix in organic chemistry, the 1989 recommendations of the Joint Commission on Biochemical Nomenclature discourage the application of the prefix "keto" for steroid names, and favor the prefix "oxo" (e.g., 11-oxo steroids rather than 11-keto steroids), because "keto" includes the carbon that is part of the steroid nucleus and the same carbon atom should not be specified twice.[36][14]
Species distribution
Steroids are found in all domains of life including bacteria, archaea, and eukaryotes. In eukaryotes, steroids are found in fungi, plants, and animals.[37][38]
Prokaryotic
In prokaryotes, biosynthetic pathways exist for the tetracyclic steroid framework (e.g. in myxobacteria)[39] – where its origin from eukaryotes is conjectured[40] – and the more-common pentacyclic triterpinoid hopanoid framework.[41]
Fungal
Fungal steroids include the ergosterols, which are involved in maintaining the integrity of the fungal cellular membrane. Various antifungal drugs, such as amphotericin B and azole antifungals, utilize this information to kill pathogenic fungi.[42] Fungi can alter their ergosterol content (e.g. through loss of function mutations in the enzymes ERG3 or ERG6, inducing depletion of ergosterol, or mutations that decrease the ergosterol content) to develop resistance to drugs that target ergosterol.[43]
Ergosterol is analogous to the cholesterol found in the cellular membranes of animals (including humans), or the phytosterols found in the cellular membranes of plants.[43] All mushrooms contain large quantities of ergosterol, in the range of tens to hundreds of milligrams per 100 grams of dry weight.[43] Oxygen is necessary for the synthesis of ergosterol in fungi.[43]
Ergosterol is responsible for the vitamin D content found in mushrooms; ergosterol is chemically converted into provitamin D2 by exposure to ultraviolet light.[43] Provitamin D2 spontaneously forms vitamin D2.[43] However, not all fungi utilize ergosterol in their cellular membranes; for example, the pathogenic fungal species Pneumocystis jirovecii does not, which has important clinical implications (given the mechanism of action of many antifungal drugs). Using the fungus Saccharomyces cerevisiae as an example, other major steroids include ergosta‐5,7,22,24(28)‐tetraen‐3β‐ol, zymosterol, and lanosterol. S. cerevisiae utilizes 5,6‐dihydroergosterol in place of ergosterol in its cell membrane.[43]
Plant
Plant steroids include steroidal alkaloids found in Solanaceae[44] and Melanthiaceae (specially the genus Veratrum),[45] cardiac glycosides,[46] the phytosterols and the brassinosteroids (which include several plant hormones).
Animal
Animal steroids include compounds of vertebrate and insect origin, the latter including ecdysteroids such as ecdysterone (controlling molting in some species). Vertebrate examples include the steroid hormones and cholesterol; the latter is a structural component of cell membranes that helps determine the fluidity of cell membranes and is a principal constituent of plaque (implicated in atherosclerosis). Steroid hormones include:
- Sex hormones, which influence sex differences and support reproduction. These include androgens, estrogens, and progestogens.
- Corticosteroids, including most synthetic steroid drugs, with natural product classes the glucocorticoids (which regulate many aspects of metabolism and immune function) and the mineralocorticoids (which help maintain blood volume and control renal excretion of electrolytes)
- Anabolic steroids, natural and synthetic, which interact with androgen receptors to increase muscle and bone synthesis. In popular use, the term "steroids" often refers to anabolic steroids.
Types
By function
The major classes of steroid hormones, with prominent members and examples of related functions, are:[47][48]
- Corticosteroids:
- Glucocorticoids:
- Cortisol, a glucocorticoid whose functions include immunosuppression
- Mineralocorticoids:
- Aldosterone, a mineralocorticoid that helps regulate blood pressure through water and electrolyte balance
- Glucocorticoids:
- Sex steroids:
- Progestogens:
- Progesterone, which regulates cyclical changes in the endometrium of the uterus and maintains a pregnancy
- Androgens:
- Testosterone, which contributes to the development and maintenance of male secondary sex characteristics
- Estrogens:
- Estradiol, which contributes to the development and maintenance of female secondary sex characteristics
- Progestogens:
Additional classes of steroids include:
- Neurosteroids such as DHEA and allopregnanolone
- Bile acids such as taurocholic acid
- Aminosteroid neuromuscular blocking agents (mainly synthetic) such as pancuronium bromide
- Steroidal antiandrogens (mainly synthetic) such as cyproterone acetate
- Steroidogenesis inhibitors (mainly exogenous) such as alfatradiol
- Membrane sterols such as cholesterol, ergosterol, and various phytosterols
- Toxins such as steroidal saponins and cardenolides/cardiac glycosides
As well as the following class of secosteroids (open-ring steroids):
- Vitamin D forms such as ergocalciferol, cholecalciferol, and calcitriol
By structure
Intact ring system
Steroids can be classified based on their chemical composition.[49] One example of how MeSH performs this classification is available at the Wikipedia MeSH catalog. Examples of this classification include:

| Class | Example | Number of carbon atoms |
|---|---|---|
| Cholestanes | Cholesterol | 27 |
| Cholanes | Cholic acid | 24 |
| Pregnanes | Progesterone | 21 |
| Androstanes | Testosterone | 19 |
| Estranes | Estradiol | 18 |
In biology, it is common to name the above steroid classes by the number of carbon atoms present when referring to hormones: C18-steroids for the estranes (mostly estrogens), C19-steroids for the androstanes (mostly androgens), and C21-steroids for the pregnanes (mostly corticosteroids).[50] The classification "17-ketosteroid" is also important in medicine.
The gonane (steroid nucleus) is the parent 17-carbon tetracyclic hydrocarbon molecule with no alkyl sidechains.[51]
Cleaved, contracted, and expanded rings
Secosteroids (Latin seco, "to cut") are a subclass of steroidal compounds resulting, biosynthetically or conceptually, from scission (cleavage) of parent steroid rings (generally one of the four). Major secosteroid subclasses are defined by the steroid carbon atoms where this scission has taken place. For instance, the prototypical secosteroid cholecalciferol, vitamin D3 (shown), is in the 9,10-secosteroid subclass and derives from the cleavage of carbon atoms C-9 and C-10 of the steroid B-ring; 5,6-secosteroids and 13,14-steroids are similar.[52]
Norsteroids (nor-, L. norma; "normal" in chemistry, indicating carbon removal)[53] and homosteroids (homo-, Greek homos; "same", indicating carbon addition) are structural subclasses of steroids formed from biosynthetic steps. The former involves enzymic ring expansion-contraction reactions, and the latter is accomplished (biomimetically) or (more frequently) through ring closures of acyclic precursors with more (or fewer) ring atoms than the parent steroid framework.[54]
Combinations of these ring alterations are known in nature. For instance, ewes who graze on corn lily ingest cyclopamine (shown) and veratramine, two of a sub-family of steroids where the C- and D-rings are contracted and expanded respectively via a biosynthetic migration of the original C-13 atom. Ingestion of these C-nor-D-homosteroids results in birth defects in lambs: cyclopia from cyclopamine and leg deformity from veratramine.[55] A further C-nor-D-homosteroid (nakiterpiosin) is excreted by Okinawan cyanobacteriosponges. e.g., Terpios hoshinota, leading to coral mortality from black coral disease.[56] Nakiterpiosin-type steroids are active against the signaling pathway involving the smoothened and hedgehog proteins, a pathway which is hyperactive in a number of cancers.[citation needed]
Biological significance
Steroids and their metabolites often function as signalling molecules (the most notable examples are steroid hormones), and steroids and phospholipids are components of cell membranes.[57] Steroids such as cholesterol decrease membrane fluidity.[58]Similar to lipids, steroids are highly concentrated energy stores. However, they are not typically sources of energy; in mammals, they are normally metabolized and excreted.
Steroids play critical roles in a number of disorders, including malignancies like prostate cancer, where steroid production inside and outside the tumour promotes cancer cell aggressiveness.[59]
Biosynthesis and metabolism
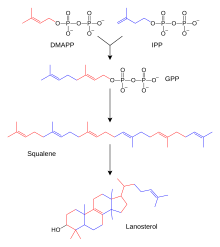
The hundreds of steroids found in animals, fungi, and plants are made from lanosterol (in animals and fungi; see examples above) or cycloartenol (in other eukaryotes). Both lanosterol and cycloartenol derive from cyclization of the triterpenoid squalene.[4] Lanosterol and cycloartenol are sometimes called protosterols because they serve as the starting compounds for all other steroids.
Steroid biosynthesis is an anabolic pathway which produces steroids from simple precursors. A unique biosynthetic pathway is followed in animals (compared to many other organisms), making the pathway a common target for antibiotics and other anti-infection drugs. Steroid metabolism in humans is also the target of cholesterol-lowering drugs, such as statins. In humans and other animals the biosynthesis of steroids follows the mevalonate pathway, which uses acetyl-CoA as building blocks for dimethylallyl diphosphate (DMAPP) and isopentenyl diphosphate (IPP).[60][better source needed]
In subsequent steps DMAPP and IPP conjugate to form farnesyl diphosphate (FPP), which further conjugates with each other to form the linear triterpenoid squalene. Squalene biosynthesis is catalyzed by squalene synthase, which belongs to the squalene/phytoene synthase family. Subsequent epoxidation and cyclization of squalene generate lanosterol, which is the starting point for additional modifications into other steroids (steroidogenesis).[61] In other eukaryotes, the cyclization product of epoxidized squalene (oxidosqualene) is cycloartenol.
Mevalonate pathway

The mevalonate pathway (also called HMG-CoA reductase pathway) begins with acetyl-CoA and ends with dimethylallyl diphosphate (DMAPP) and isopentenyl diphosphate (IPP).
DMAPP and IPP donate isoprene units, which are assembled and modified to form terpenes and isoprenoids[62] (a large class of lipids, which include the carotenoids and form the largest class of plant natural products).[63] Here, the isoprene units are joined to make squalene and folded into a set of rings to make lanosterol.[64] Lanosterol can then be converted into other steroids, such as cholesterol and ergosterol.[64][65]
Two classes of drugs target the mevalonate pathway: statins (like rosuvastatin), which are used to reduce elevated cholesterol levels,[66] and bisphosphonates (like zoledronate), which are used to treat a number of bone-degenerative diseases.[67]
Steroidogenesis
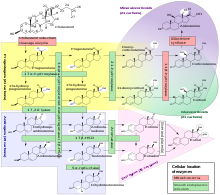
Steroidogenesis is the biological process by which steroids are generated from cholesterol and changed into other steroids.[69] The pathways of steroidogenesis differ among species. The major classes of steroid hormones, as noted above (with their prominent members and functions), are the progestogens, corticosteroids (corticoids), androgens, and estrogens.[25][70] Human steroidogenesis of these classes occurs in a number of locations:
- Progestogens are the precursors of all other human steroids, and all human tissues which produce steroids must first convert cholesterol to pregnenolone. This conversion is the rate-limiting step of steroid synthesis, which occurs inside the mitochondrion of the respective tissue.[71][25][better source needed]
- Cortisol, corticosterone, aldosterone are produced in the adrenal cortex.[25][70]
- Estradiol, estrone and progesterone are made primarily in the ovary, estriol in placenta during pregnancy, and testosterone primarily in the testes[25][72][73][74] (some testosterone may also be produced in the adrenal cortex).[25][70]
- Estradiol is converted from testosterone directly (in males), or via the primary pathway DHEA – androstenedione – estrone and secondarily via testosterone (in females).[25]
- Stromal cells have been shown to produce steroids in response to signaling produced by androgen-starved prostate cancer cells.[59][non-primary source needed][better source needed]
- Some neurons and glia in the central nervous system (CNS) express the enzymes required for the local synthesis of pregnenolone, progesterone, DHEA and DHEAS, de novo or from peripheral sources.[25][citation needed]
| Sex | Sex hormone | Reproductive phase | Blood production rate | Gonadal secretion rate | Metabolic clearance rate | Reference range (serum levels) | |
|---|---|---|---|---|---|---|---|
| SI units | Non-SI units | ||||||
| Men | Androstenedione | – | 2.8 mg/day | 1.6 mg/day | 2200 L/day | 2.8–7.3 nmol/L | 80–210 ng/dL |
| Testosterone | – | 6.5 mg/day | 6.2 mg/day | 950 L/day | 6.9–34.7 nmol/L | 200–1000 ng/dL | |
| Estrone | – | 150 μg/day | 110 μg/day | 2050 L/day | 37–250 pmol/L | 10–70 pg/mL | |
| Estradiol | – | 60 μg/day | 50 μg/day | 1600 L/day | <37–210 pmol/L | 10–57 pg/mL | |
| Estrone sulfate | – | 80 μg/day | Insignificant | 167 L/day | 600–2500 pmol/L | 200–900 pg/mL | |
| Women | Androstenedione | – | 3.2 mg/day | 2.8 mg/day | 2000 L/day | 3.1–12.2 nmol/L | 89–350 ng/dL |
| Testosterone | – | 190 μg/day | 60 μg/day | 500 L/day | 0.7–2.8 nmol/L | 20–81 ng/dL | |
| Estrone | Follicular phase | 110 μg/day | 80 μg/day | 2200 L/day | 110–400 pmol/L | 30–110 pg/mL | |
| Luteal phase | 260 μg/day | 150 μg/day | 2200 L/day | 310–660 pmol/L | 80–180 pg/mL | ||
| Postmenopause | 40 μg/day | Insignificant | 1610 L/day | 22–230 pmol/L | 6–60 pg/mL | ||
| Estradiol | Follicular phase | 90 μg/day | 80 μg/day | 1200 L/day | <37–360 pmol/L | 10–98 pg/mL | |
| Luteal phase | 250 μg/day | 240 μg/day | 1200 L/day | 699–1250 pmol/L | 190–341 pg/mL | ||
| Postmenopause | 6 μg/day | Insignificant | 910 L/day | <37–140 pmol/L | 10–38 pg/mL | ||
| Estrone sulfate | Follicular phase | 100 μg/day | Insignificant | 146 L/day | 700–3600 pmol/L | 250–1300 pg/mL | |
| Luteal phase | 180 μg/day | Insignificant | 146 L/day | 1100–7300 pmol/L | 400–2600 pg/mL | ||
| Progesterone | Follicular phase | 2 mg/day | 1.7 mg/day | 2100 L/day | 0.3–3 nmol/L | 0.1–0.9 ng/mL | |
| Luteal phase | 25 mg/day | 24 mg/day | 2100 L/day | 19–45 nmol/L | 6–14 ng/mL | ||
Notes and sources Notes: "The concentration of a steroid in the circulation is determined by the rate at which it is secreted from glands, the rate of metabolism of precursor or prehormones into the steroid, and the rate at which it is extracted by tissues and metabolized. The secretion rate of a steroid refers to the total secretion of the compound from a gland per unit time. Secretion rates have been assessed by sampling the venous effluent from a gland over time and subtracting out the arterial and peripheral venous hormone concentration. The metabolic clearance rate of a steroid is defined as the volume of blood that has been completely cleared of the hormone per unit time. The production rate of a steroid hormone refers to entry into the blood of the compound from all possible sources, including secretion from glands and conversion of prohormones into the steroid of interest. At steady state, the amount of hormone entering the blood from all sources will be equal to the rate at which it is being cleared (metabolic clearance rate) multiplied by blood concentration (production rate = metabolic clearance rate × concentration). If there is little contribution of prohormone metabolism to the circulating pool of steroid, then the production rate will approximate the secretion rate." Sources: See template. | |||||||
Alternative pathways
In plants and bacteria, the non-mevalonate pathway (MEP pathway) uses pyruvate and glyceraldehyde 3-phosphate as substrates to produce IPP and DMAPP.[62][75]
During diseases pathways otherwise not significant in healthy humans can become utilized. For example, in one form of congenital adrenal hyperplasia a deficiency in the 21-hydroxylase enzymatic pathway leads to an excess of 17α-Hydroxyprogesterone (17-OHP) – this pathological excess of 17-OHP in turn may be converted to dihydrotestosterone (DHT, a potent androgen) through among others 17,20 Lyase (a member of the cytochrome P450 family of enzymes), 5α-Reductase and 3α-Hydroxysteroid dehydrogenase.[76]
Catabolism and excretion
Steroids are primarily oxidized by cytochrome P450 oxidase enzymes, such as CYP3A4. These reactions introduce oxygen into the steroid ring, allowing the cholesterol to be broken up by other enzymes into bile acids.[77] These acids can then be eliminated by secretion from the liver in bile.[78] The expression of the oxidase gene can be upregulated by the steroid sensor PXR when there is a high blood concentration of steroids.[79] Steroid hormones, lacking the side chain of cholesterol and bile acids, are typically hydroxylated at various ring positions or oxidized at the 17 position, conjugated with sulfate or glucuronic acid and excreted in the urine.[80]
Isolation, structure determination, and methods of analysis
Steroid isolation, depending on context, is the isolation of chemical matter required for chemical structure elucidation, derivitzation or degradation chemistry, biological testing, and other research needs (generally milligrams to grams, but often more[81] or the isolation of "analytical quantities" of the substance of interest (where the focus is on identifying and quantifying the substance (for example, in biological tissue or fluid). The amount isolated depends on the analytical method, but is generally less than one microgram.[82][page needed]
The methods of isolation to achieve the two scales of product are distinct, but include extraction, precipitation, adsorption, chromatography, and crystallization. In both cases, the isolated substance is purified to chemical homogeneity; combined separation and analytical methods, such as LC-MS, are chosen to be "orthogonal"—achieving their separations based on distinct modes of interaction between substance and isolating matrix—to detect a single species in the pure sample.
Structure determination refers to the methods to determine the chemical structure of an isolated pure steroid, using an evolving array of chemical and physical methods which have included NMR and small-molecule crystallography.[2]: 10–19 Methods of analysis overlap both of the above areas, emphasizing analytical methods to determining if a steroid is present in a mixture and determining its quantity.[82]
Chemical synthesis
Microbial catabolism of phytosterol side chains yields C-19 steroids, C-22 steroids, and 17-ketosteroids (i.e. precursors to adrenocortical hormones and contraceptives).[83][84][85] The addition and modification of functional groups is key when producing the wide variety of medications available within this chemical classification. These modifications are performed using conventional organic synthesis and/or biotransformation techniques.[86][87]
Precursors
Semisynthesis
The semisynthesis of steroids often begins from precursors such as cholesterol,[85] phytosterols,[84] or sapogenins.[88] The efforts of Syntex, a company involved in the Mexican barbasco trade, used Dioscorea mexicana to produce the sapogenin diosgenin in the early days of the synthetic steroid pharmaceutical industry.[81]
Total synthesis
Some steroidal hormones are economically obtained only by total synthesis from petrochemicals (e.g. 13-alkyl steroids).[85] For example, the pharmaceutical Norgestrel begins from methoxy-1-tetralone, a petrochemical derived from phenol.
Research awards
A number of Nobel Prizes have been awarded for steroid research, including:
- 1927 (Chemistry) Heinrich Otto Wieland — Constitution of bile acids and sterols and their connection to vitamins[89]
- 1928 (Chemistry) Adolf Otto Reinhold Windaus — Constitution of sterols and their connection to vitamins[90]
- 1939 (Chemistry) Adolf Butenandt and Leopold Ružička — Isolation and structural studies of steroid sex hormones, and related studies on higher terpenes[91]
- 1950 (Physiology or Medicine) Edward Calvin Kendall, Tadeus Reichstein, and Philip Hench — Structure and biological effects of adrenal hormones[92]
- 1965 (Chemistry) Robert Burns Woodward — In part, for the synthesis of cholesterol, cortisone, and lanosterol[93]
- 1969 (Chemistry) Derek Barton and Odd Hassel — Development of the concept of conformation in chemistry, emphasizing the steroid nucleus[94]
- 1975 (Chemistry) Vladimir Prelog — In part, for developing methods to determine the stereochemical course of cholesterol biosynthesis from mevalonic acid via squalene[95]